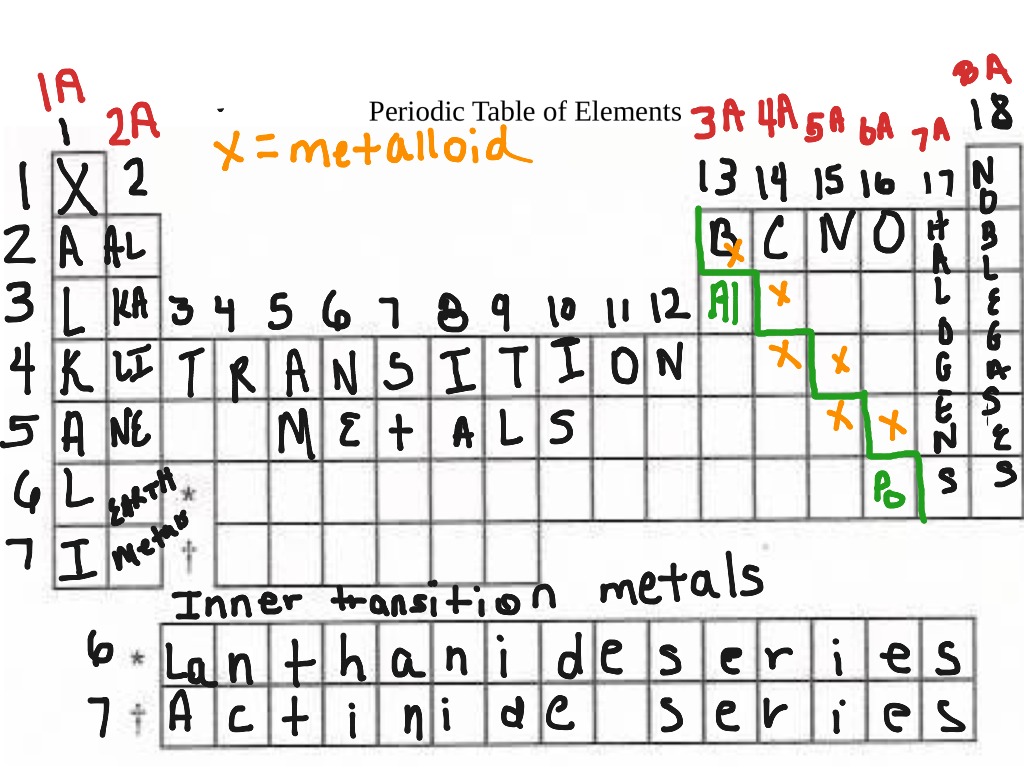
Figure 2.27: The periodic table organizes elements with similar properties into groups. We can sort the elements into large classes with common properties: metals (elements that are shiny, malleable, good conductors of heat and electricity-shaded yellow) nonmetals(elements that appear dull, poor conductors of heat and electricity-shaded green) and metalloids(elements that conduct heat and electricity moderately well, and possess some properties of metals and some properties of nonmetals-shaded purple). Figure 2.26: Elements in the periodic table are organized according to their properties. For the table to fit on a single page, parts of two of the rows, a total of 14 columns, are usually written below the main body of the table. IUPAC recommends that the numbers 1 through 18 be used, and thus this labelling is more common. Groups are labelled at the top of each column. The elements are arranged in seven horizontal rows, called periods or series, and 18 vertical columns, called groups. Each box represents an element and contains its atomic number, symbol, average atomic mass, and (sometimes) name. The modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column (Figure 2.26).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
